Refeeding Syndrome (RS) is a complex clinical picture that occurs as a consequence of the reintroduction of nutrition (oral, enteral or parenteral) in malnourished patients.
Patients present with fluid balance disorders, electrolyte abnormalities – such as hypophosphatemia, hypokalemia, and hypomagnesemia – alterations in carbohydrate metabolism, and vitamin deficiencies.
This results in the appearance of neurological, respiratory, cardiac, neuromuscular and hematological complications.
In this article the pathogenesis and clinical characteristics of RS have been reviewed, making some suggestions for its prevention and treatment.
The most important thing in the prevention of RS is to identify patients at risk, establish nutritional support in a prudent way and perform an adequate correction of electrolyte and vitamin deficiencies.
Definition
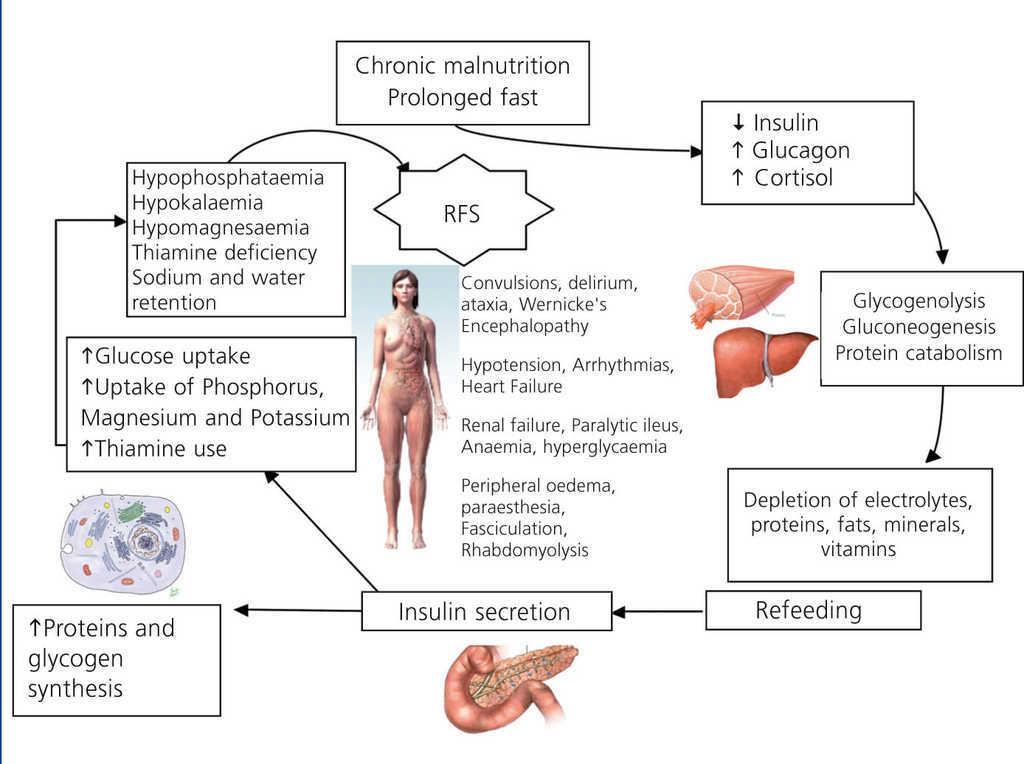
The fundamental fact of Refeeding Syndrome is severe hypophosphatemia2, which is accompanied by abnormalities in fluid balance, alterations in carbohydrate metabolism and certain vitamin deficiencies, for example in thiamine, as well as hypokalemia and hypomagnesemia2.
This translates clinically into the appearance of, among others, neurological, respiratory, cardiovascular and hematological alterations a few days after the start of refeeding, which leads to increased morbidity and even mortality in the patient3.
The classic study describing RS was conducted by Keys et al4 in healthy male conscientious objectors from World War II. Participants were semi-fasted for 6 months, after which a normal oral diet was reintroduced.
The consequence was a decrease in the cardiovascular reserve, with heart failure in some cases. Similar clinical results could be seen when reinstating a normal intake in individuals who had suffered siege or had been interned in concentration camps in World War II5.
Later, with the appearance of parenteral (PN) and enteral (EN) nutrition, the same type of complications was observed in malnourished patients who received aggressive nutritional support6.
In addition to RS, especially in seriously ill hospitalized patients, there are numerous causes of hypophosphatemia, hypomagnesemia, and hypokalemia. The most common should be kept in mind when making the differential diagnosis.
Hypophosphatemia is a relatively common finding in hospitalized patients; it affects 3-42% of them. The incidence is especially high in intensive care and infectious disease units10.
Marik and Bedigian11 detected feedback-related hypophosphatemia in 34% of intensive care patients after a fast of just 48 hours.
Pathophysiology
Tissues preferentially use carbohydrates for energy. Our body has a limited reserve, in the form of glycogen stored in the liver and muscle.
During the initial period of fasting, glycogen stores are used as an energy source.
After the depletion of these deposits, proteolysis begins, which supplies amino acids for gluconeogenesis, which will allow glucose-dependent tissues (brain, kidney marrow, red cells) to have glucose.
After 72 h of fasting and in order to avoid the mobilization of skeletal muscle proteins, the metabolic pathways lead to lipolysis, obtaining free fatty acids.
These free fatty acids can follow two routes: at the peripheral level they are used by cells to produce energy, and at the liver level they are used as substrates for the synthesis of ketone bodies (acetoacetic acid, β-hydroxybutyric acid and acetone) through ketogenesis.
The brain can use ketone bodies, which cross the blood-brain barrier, as an energy source.
However, the ability of cells to oxidize ketone bodies is limited, leading to ketosis and ultimately metabolic acidosis.
All this hormonal change leads to a decrease in basal metabolism and increases the availability of certain energy substrates.
During fasting, in addition to weight loss, there is a decrease in cell mass and an increase in extracellular water.
Plasma levels of electrolytes such as potassium, phosphorus and magnesium remain within normal limits; however, your total body content is decreased.
The pathogenesis of RS is complex since metabolic and physiological changes intervene during the phase of depletion and repletion of substrates, with the consequent compartmental deviations of electrolytes, alterations in the metabolism of glucose and vitamins and in the management of body water .
The exact mechanism by which fluid imbalance occurs is unknown, but it is believed that sodium and water retention may be due to an antinatriuretic effect of hyperinsulinemia16.
Thiamine is an essential cofactor in the metabolism of carbohydrates17, therefore, the contribution of a high quantity of these increases its demand.
Although it is difficult to establish whether thiamine deficiency is due to refeeding or the fasting state, malnourished patients are at risk of developing deficiency and its associated complications.
There is also an increase in the conversion of T4 to T318,19, which causes an increase in energy expenditure. Figure 1 shows the pathophysiology of SR.